1. The role of dual-specificity phosphatases in MAP kinase signaling pathways.
The family of mitogen-activated protein kinases (MAPK) includes the c-Jun N-terminal protein kinases (JNK) that are activated by pro-inflammatory cytokines, mitogenic stimuli and a variety of environmental stresses. These kinases are key regulators of the activities of many transcription factors, which include c-Jun, ATF2 and Elk1, controlling diverse gene expression programs. The magnitude and duration of activation of this pathway may lead to entirely opposite effects in the cell, i.e. survival or apoptosis. Therefore, as well as being regulated positively by phosphorylation, regulation of MAPKs at the level of dephosphorylation is thought to be very important in determining specific signaling outcomes. It has been shown that exposure of cells to protein damaging stresses, including arsenite, oxidative stress and heat shock, strongly reduces the rate of JNK dephosphorylation. Regulation of the JNK pathway by arsenite may be relevant in carcinogenesis since chronic arsenic exposure is associated with increased risk of human cancer of the skin, bladder, kidney, lung and colon. A dual-specificity phosphatase (DUSP8, also called M3/6) has been identified, showing specificity primarily towards JNK and p38. The activity of this phosphatase is inhibited by arsenite, thus leading to an increase in JNK phosphorylation and activity. We are investigating the exact mechanisms of regulation of DUSP8 activity, aiming for a better understanding of the role of this enzyme in cell physiology and disease processes. Specific lines of research include:
- Application of mass spectrometry to identify post-translational modifications on DUSP8.
- Characterization of the regulatory role of specific DUSP8 sites phosphorylated by JNK.
- Specificity of DUSP8 interactions with distinct JNK isoforms and JIP scaffold proteins.
- Identification of novel DUSP8-interacting proteins by affinity purification and mass spectroscopy.
- Investigation of the mechanisms regulating DUSP8 activity upon arsenite induced oxidative stress.
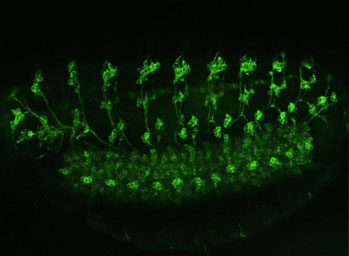
- Study of the in vivo role of the JNK-specific DUSP Puckered using Drosophila as a model system.
- Construction of transgenic mice expressing active or dominant-negative forms of DUSP8 in order to analyse its role in disease models.
- Construction of DUSP8-null mice using CRISPR/Cas9 methodology and investigation of gene expression and proteomic profiles of mutant mouse tissues.
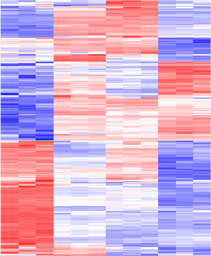
2. Proteomic approaches for the study of oncogenic signaling.
Recent advances in proteomic technologies have enabled their application in understanding the complex responses of cells to tumorigenic transformation. We employ various cellular systems that model the oncogenic subversion of signaling pathways found in many human cancers. Proteomic methodologies are used to identify differences in protein expression or post-translational modification that correlate with the transformation process. Selected proteins are then validated with other methods and analysed further in order to understand mechanisms of oncogenic transformation. Examples of model systems used include:
-
Colon cancer cells expressing mutant, activated forms of Ras oncogenes.
-
B-lymphocytes transformed by Eppstein-Barr virus oncogenes.
-
Breast cancer cells expressing high levels of the oncogenic receptor tyrosine-kinase HER2.